Lake Wawasee: how temperature affects the lake year-round
This post has been updated to include the completed, year-round study on Lake Wawasee. Download the report today.
Two long-standing Lake Wawasee families, the Herdrichs and the Griffiths, funded a year-long monthly sampling effort on the lake to see what’s going on under the surface in the off-season. Let’s take a look at what we saw!
A lake’s temperature impacts many aspects of the lake’s ecosystem, including dissolved oxygen and clarity.
During the summer, the topmost level of Wawasee is warm and the bottom layer is cold. At some point during autumn, these layers “turnover,” pushing surface water downward and oxygen-starved bottom water toward the surface. And as it turns out, the winter can be a time of rejuvenation for a lake!
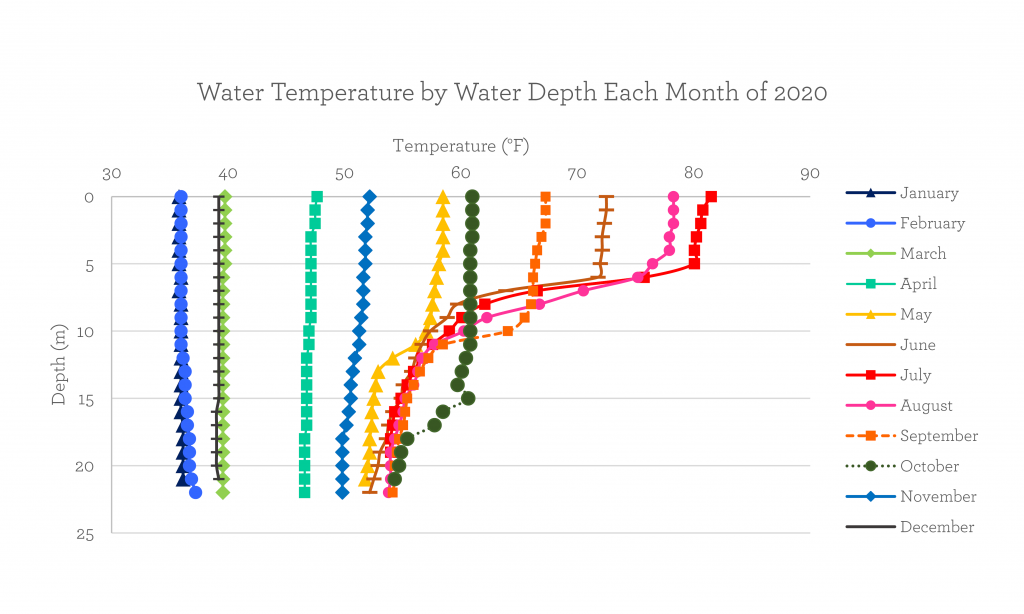
How cold is the lake in the wintertime?
If we analyze the temperature figure above, we see that the first and last few months of 2020 consist of straight lines.
This shows that the lake is the same temperature top to bottom, and that temperature can be pretty cold! Our January sampling, which occurred before the lake froze over, showed the lake was 36°F. That’s colder than your fridge but not as cold as your freezer.
The lake froze over fully on January 20, 2020 (also known as the ice-on date), and thawed on March 6 (ice-off). You may be able to predict when the lake will freeze based on the water’s appearance; as the water temperature gets closer to freezing, the surface can look heavier or syrupy. Much like syrup, it seems “thicker” in this very cold, dense state. Too much colder, and the water will start to expand and form crystals: ice!
You may have also noticed that the lines in the graph bend heavily in the summer months. This is when the lake is stratified: differences in temperature separate the lake into three non-mixing layers.
The months where the temperature line starts to straighten out again show us lake turnover is taking place. Lake turnover is the mixing of all three lake layers as surface temperatures become less extreme, and in Wawasee (as with all of our other lakes) it usually occurs twice a year: once in the spring and once in the autumn. In 2020, turnover occurred in April and November.
This cycle acts as an ecological reset button, creating a healthier balance throughout the entire lake by refreshing dissolved oxygen levels in the bottom water. That’s good news for fish and other aquatic critters who rely on oxygen!
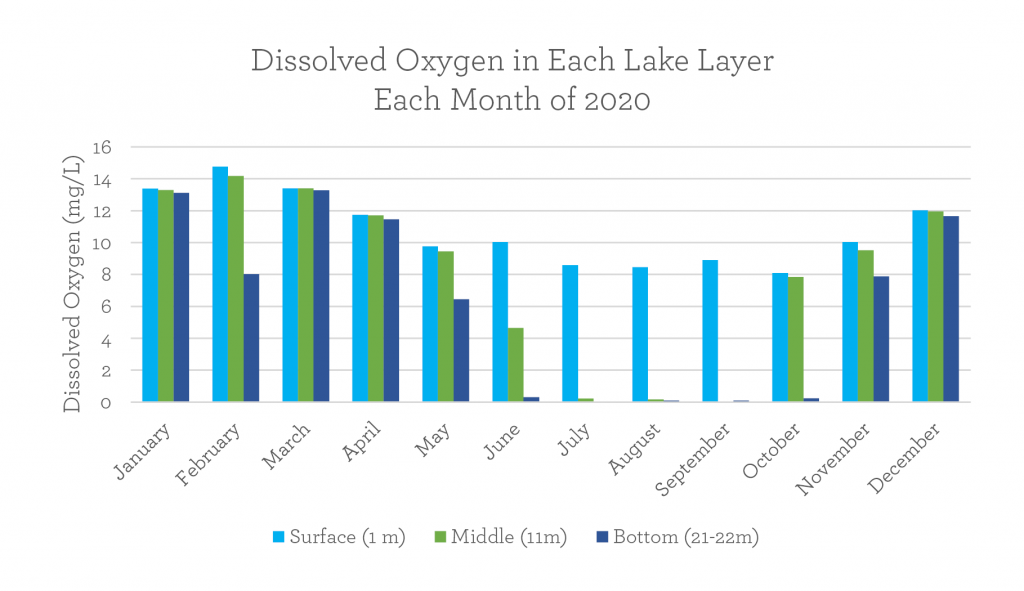
How do oxygen levels change through the seasons?
Freshwater organisms such as fish, bacteria or macroinvertebrates need dissolved oxygen (DO) in order to breathe.
DO near the surface is abundant, ranging from 8.1 – 14.8 mg/L on Wawasee last year. But DO at the lake bottom varies much more; it ranged from 0.1 – 13.8 mg/L last year. For reference, most fish need more than 2.0 mg/L of DO to survive! Take a look at the bar graph above. You’ll notice that the late fall to early spring show consistently higher amounts of DO across all lake layers. Meanwhile, from late spring to mid-autumn, DO tends to drop.
Remember that the effects of lake turnover restores the balance dissolved oxygen through the whole lake! In December and March, during and after turnover, all layers of lake Wawasee contained nearly equal amounts of DO.
During summer stratification, however, DO cannot flow between layers.
DO concentrations in the bottom layers plummet as normal consumption and decomposition happens, and by mid-summer, nearly all the DO in the bottom half of the lake is gone. This depletion isn’t as extreme in the winter months because the colder temperatures reduce the intensity of the lake’s stratification. This allows DO to move throughout all parts of the lake. Plus, oxygen-using organisms tend to be less active in cooler temperatures!
That being said, if winter hangs around too long and the ice stays in a thick layer on the surface of a lake, dissolved oxygen will eventually begin to decrease at the bottom of the lake, like we see at the bottom of the February line on this figure. Fresh oxygen gets mixed into lakes with wind, and that can only happen when there’s no ice! Spring turnover in March quickly restores DO in preparation for summertime.
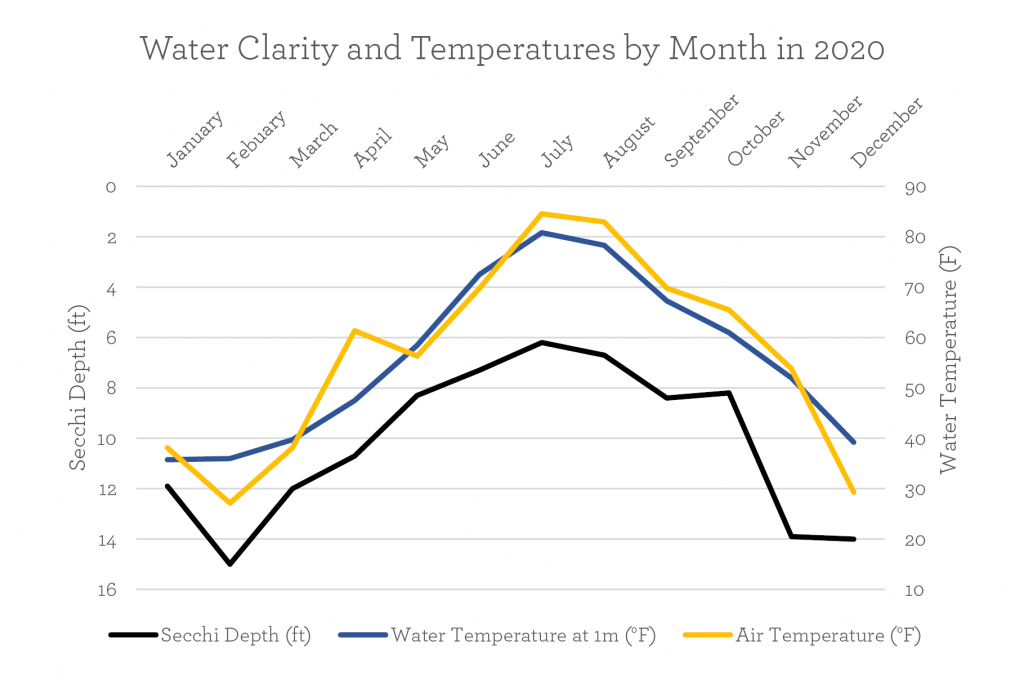
Water clarity is measured by a Secchi disk, which is lowered into the water until the white and black are indistinguishable. The depth of disappearance is recorded.
From our sampling, we learned that the Wawasee Secchi depth is actually very deep during winter months (shown by the low black line on the figure above). The summer has shallow water clarity (the “hill” in the middle of the black line). In these monthly 2020 samples, Wawasee’s water clarity varied from 6.2 ft to 15.0 ft into the water. In the figure, the Secchi depth line follows the water and air temperature lines, with warmer months having less clear water.
Does this mean that colder water is more clear than warm water? Not necessarily!
It’s not the cold temperature that gives winter water greater clarity, but the effects that the cold water has on what lives in a lake’s ecosystem, such as algae. Algae, other microorganisms, and sediment are what determine how murky or cloudy lake water appears. Since algae are sort of like microscopic plants, they thrive best in warmer, sunnier conditions where they can photosynthesize and make energy.
Also during the summer, Wawasee is constantly in use. Boats and other watercraft can stir up sediment on the bottom of the lake, making the water more cloudy and inspiring algae growth with nutrients. Shorelines are also more likely to erode due to seasonal rain and wakes hitting the shore. You might notice a similar effect when you shake a snowglobe: there are lots of little particles that get thrown higher into the water, and those particles won’t settle down to the bottom until the movement stops.
But no matter what time of year you’re admiring your lake, there’s always something happening under the surface!
Even in the cold stillness of winter, lakes undergo change that can impact the whole rest of the year. This monthly sampling project on Lake Wawasee helped us see some of those changes in just a few key measurements: temperature, dissolved oxygen, and water clarity.

Download the full study
Curious about our sampling methods or want to see all of our results? Review the data for yourself today.